 
There are three ways of arranging two substituents around a benzene ring, meaning that disubstituted rings have three structural isomers, hence the need for a discussion of structural isomers and substituted benzene rings in one article. In this installment, we expand our knowledge of the IR spectroscopy of aromatic rings by looking at the spectra of mono- and disubstituted benzene rings. In the last installment (2), I introduced the topic of aromatic bonding and aromatic rings, and we looked at the spectrum of the prototypical aromatic ring benzene in detail. However, infrared (IR) spectroscopy, where the peak positions are determined by the atomic masses and bond strengths of the functional group undergoing vibration (1), can distinguish between structural isomers because each structural isomer has a unique set of masses and bond strengths. Structural isomers have the same mass so they are difficult to distinguish by mass spectrometry. 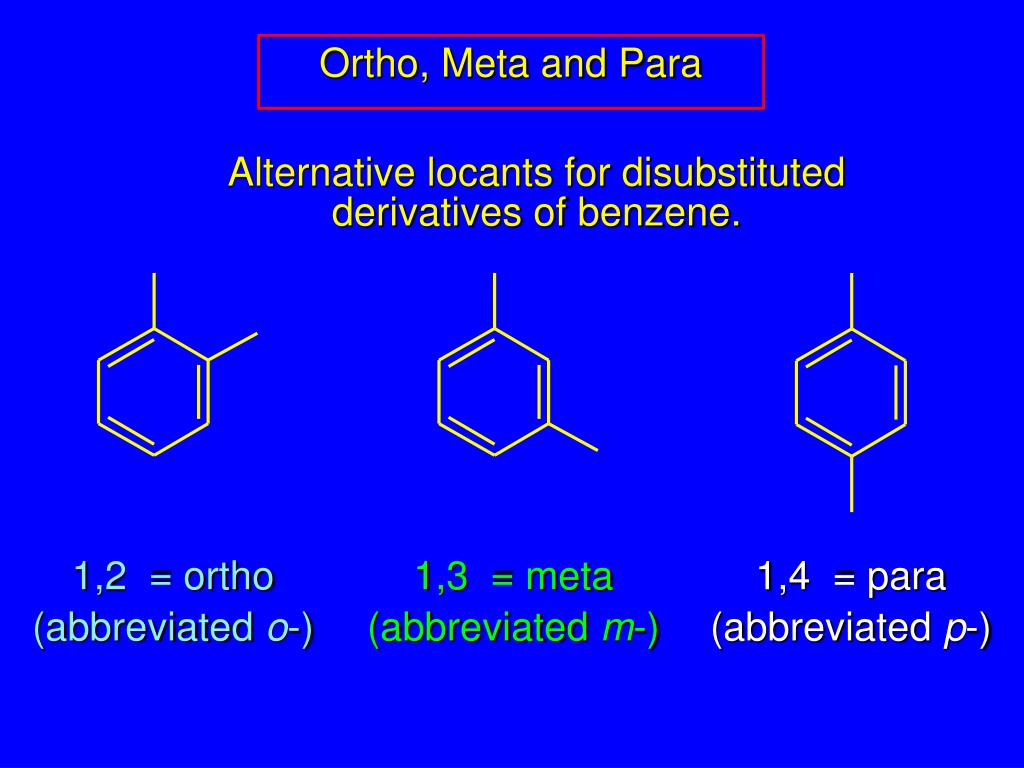
Therefore, these molecules have different chemical properties such as boiling points and reactivities. Structural isomers are molecules that have the same chemical formula but different chemical structures. We examine numerous example spectra and learn how the position of C-H wagging peaks, and the presence or absence of a ring-bending peak, allow one to distinguish between mono-, ortho-, meta-, and para-substituted rings most of the time. Following up on the last installment, we examine the infrared (IR) spectra of mono- and disubstituted benzene rings. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
